- Blog
- Ip address location reveal omegle software
- Dewalt 18v battery adapter
- Gaming software logitech g29
- Travel expense report template excel
- Sonic mania mod loader crashes game
- Free save wizard license key free
- Doterra emotional aromatherapy wheel
- Fake drivers license maker free online inages
- Budget planners templates
- Twilight forest minecraft mod download
- Spectrum tv choice channells
- Space shuttle endeavour la
- Top 5 online hentai game website
- Nigeria time zone gmt
- Editing pes 2017 kit -bin files
- Xforce keygen for autocad 2016
- Signs and symptoms of blunt force trauma
- Free product key for xplane 11
- Florida tattoo ideas cursive font
- Rome total war 2 faction tier list
- Volleyball referee signals high school
- Lord of the rings noble collection crown
- Putty ssh end session command
- K40 laserdrw 3 settings
- Tennessee keno winning numbers
- Slime rancher multiplayer mod free
- Will spectrasonics omnisphere 2 replace omnisphere 1
- Free lance invoice template microsoft excel
- Periodic table with exact molar mass
- Weekly work schedule template google free
- Hindi typing tutor kruti dev
- Tomodachi life pc version
- Bongiovi dps for windows 10 crack
- Linkedin logo linkedin icon png
- Normal xray of hip
- Weight tracker template
- Funny fantasy football team names cardinals
- Solving quadratic equations by square roots
- Optimization library not found halion
- Yearly household budget template excel


Thus, the formula mass of calcium hydrogen carbonate is 117.10 amu and the molar mass of calcium hydrogen carbonate is 117.10 grams per mole (g/mol).
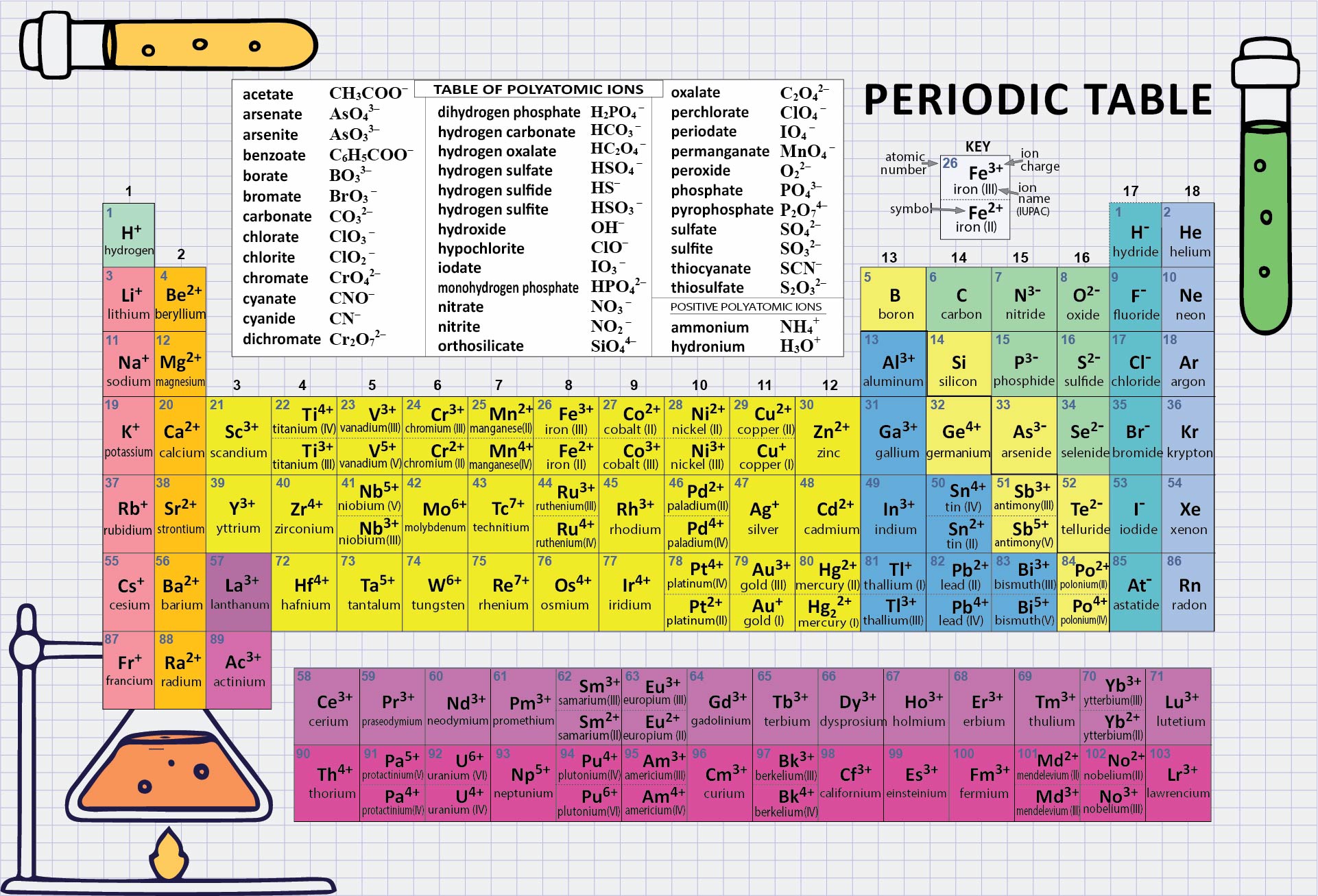
However when talking about a mole of an ionic compound we will still use the term molar mass. This is because there are no individual molecules in ionic compounds. To find the mole fraction and percentage of each element in Cr, divide each total from step 3 by the total molar mass found in step 4: Mole Fraction. For compounds that are not molecular (ionic compounds), it is improper to use the term “molecular mass” and “ formula mass” is generally substituted. Finally, add together the total mass of each element to get the molar mass of Cr: 51.9961 g/mol 51.9961 g/mol. The molar mass of the N 2 molecule is therefore 28.02 g/mol. This is referred to as the molecular mass and the molecular mass of any molecule is simply the sum of the atomic masses of all of the elements in that molecule. Meija et al in Atomic Weights of the Elements 2013, and the isotopic compositions data were published by M. The atomic weights data were published by J. For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) = 28.02 amu. The atomic weights are available for elements 1 through 118 and isotopic compositions or abundances are given when appropriate. For a molecule (for example, nitrogen, N 2) the mass of molecule is the sum of the atomic masses of the two nitrogen atoms. The concept of molar mass can also be applied to compounds. Further, if you have 16.00 grams of oxygen atoms, you know from the definition of a mole that your sample contains 6.022 x 10 23 oxygen atoms. For example, the atomic mass of an oxygen atom is 16.00 amu that means the molar mass of an oxygen atom is 16.00 g/mol. Generalizing this definition, the molar mass of any substance in grams per mole is numerically equal to the mass of that substance expressed in atomic mass units. By definition, the molar mass of carbon-12 is numerically the same, and is therefore exactly 12 grams.
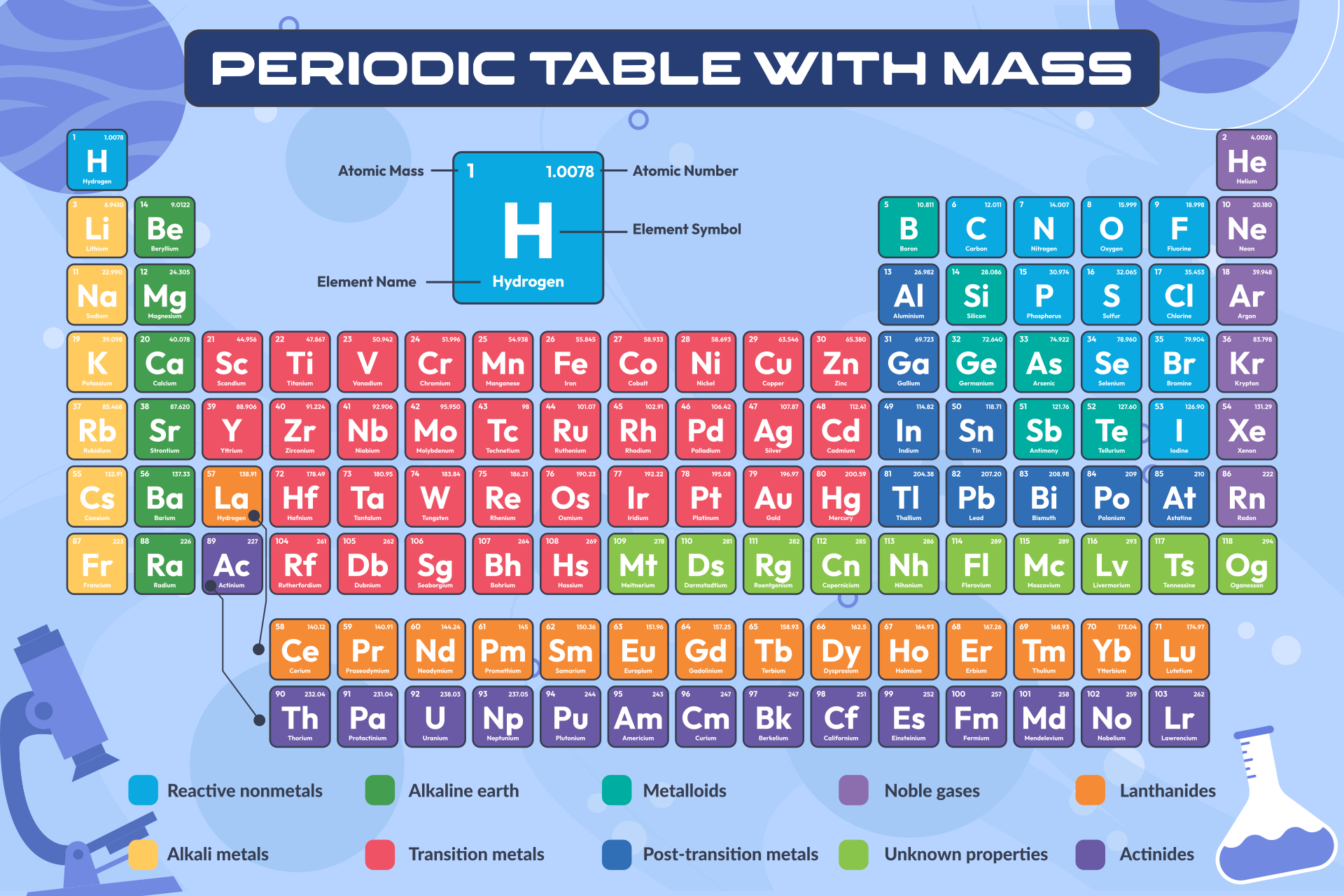
We have defined a mole based on the isotopic atomic mass of carbon-12. Use the buttons above to change your view of the periodic table and view Murray Robertson’s stunning Visual Elements artwork. Click the tabs at the top to explore each section. The atomic mass of an element is the relative average of all of the naturally occurring isotopes of that element and atomic mass is the number that appears in the periodic table. Periodic Table The Royal Society of Chemistry's interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. The molar mass of a compound is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol)īecause the definitions of both the mole and the formula mass are based on the same reference substance, 12C, the molar mass of any compound is numerically equivalent to its formula weight in amu.) a carbon atom with six proton and six neutrons in its’ nucleus, surrounded by six electrons. The masses of 1 mole of different compunds, however, are different, since the masses of the individual molecules are drastically different. Similar to atoms, 1 mole of any compound contains the same number of molecules as 1 mole of any other compund.
